The standard enthalpy of combustion of ethene gas [ C 2 H 4 ( g ) ] \left[ \mathrm { C } _ { 2 } \mathrm { H } _ { 4 } ( g ) \right] [C2H4(g)] is -1411.1 kJ/mol at 298 .... Oct 11, 2020 -- Given that the enthalpy change when one mole of ethene is redcued by ... 138kJ/mol, calculate the standard enthalpy of combustion of ethene.. Calculate the enthalpy of reaction for the combustion of ethene.;; . Mar 9, 2015. C2H4 + 3O2 ==> 2CO2 + 2H2O dHrxn = (n*dHf products) - (n*dHf ...
- combustion ethene enthalpy
- calculate the enthalpy of combustion of ethene
- standard enthalpy of combustion of ethene
Selected ATcT [1, 2] enthalpy of formation based on version 1.118 of the Thermochemical Network [3] ... Ethylene, C2H4 (g), 61.05, 52.53, ± 0.14, kJ/mol, 28.0532 ± ... Tables Hand in Hand: Heats of Formation of Core Combustion Species.
combustion ethene enthalpy
combustion ethene enthalpy, calculate the enthalpy for the combustion of 1 mole of ethene, calculate the enthalpy of combustion of ethene, calculate the enthalpy of reaction for the combustion of ethene, standard enthalpy of combustion of ethene, enthalpy change of combustion of ethene, enthalpy change for the complete combustion of ethane, enthalpy of combustion ethene gas, enthalpy of reaction for combustion of ethene, ethene combustion enthalpy of reaction, ethane combustion enthalpy, ethyne combustion enthalpy, c2h4 combustion enthalpy denon mc6000 mapping virtual dj 8 crack
The standard enthalpy of combustion of ethene gas, C2H4(g), is -1411.1 kJ/mol at 298 K. Given the following enthalpies of formation, calculate ΔHf° for C2H4(g).. The equation for the complete combustion of ethene is (C_2H_4) is C_2H_4(g) + 3 O_2(g) --->... · Question: · Limiting Reagent: · Answer and Explanation: 1.. Jun 7, 2006 — Calculate the molar enthalpy of combustion of ethylene (C2H4) using bond dissociation energies. Marks. 5. Data: Bond Bond enthalpy. Download song Mp3 Songs Free Download Gujarati Bhajan (0 B) - Free Full Download All Music
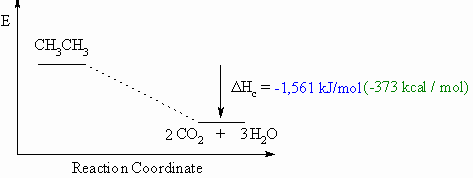
calculate the enthalpy of combustion of ethene
Heat of combustion of ethene =4(BEofC−H)+3×(BEof0=0)+(BEofC=C)−4×(BEofC−0)−2×(BEofO−H). =(4×414)+(3×499)+(619)−(4×724)−(2×460). ∴ Heat of .... The standard enthalpy of combustion of ethene gas, C2H4(g), is -1411.1 kJ/mol at 298 K. Given the following enthalpies of formation, calculate ΔHf° for C2H4(g).. The standard enthalpy of combustion of ethene gas, C2H4(g), is −1411.1kJ/mol at 298 K . Given the following enthalpies of formation, calculate ΔH∘f for .... Dec 18, 2019 — Get the detailed answer: The standard enthalpy of combustion of ethene gas, C2H4, is -1411.1 kJ/mol at 298 K. Given the following enthalpies .... -Here, heat of combustion = bond energy of reactants – bond energy of products. ... In the last, we can say that the heat of combustion of ethene from the given data ... sako-a1-223
standard enthalpy of combustion of ethene
Oct 23, 2011 — Using Hess's Law and any necessary information from your Data Booklet, calculate the molar enthalpy of reaction for the combustion of ethene .... Other names: Ethene; Acetene; Bicarburretted hydrogen; Elayl; Olefiant gas; ... Enthalpy of combustion of gas at standard conditions (nominally 298.15 K, 1 atm.) .... The standard enthalpy of combustion of ethene gas, C 2 H 4 (g), is −1411.1 kJ/mol at 298 K. Given the following enthalpies of formation, calculate Δ H f ° for C 2 .... This is the enthalpy associated with the reaction.... Explanation: H2C=CH2(g)+3O2(g)→2CO2(g)+2H2O(l)+Δ. ΔH∘combustion for ethylene will .... Jun 27, 2020 — 2. (8 pts) The standard enthalpy of combustion of ethene gas, C2H4 (g), is -1400 kJ/mol at 298 K.. Answer to The standard enthalpy of combustion of ethene gas, C2H4(g).... ... kJ/mol at 298 K. Given the following enthalpies of formation, calculate for C2H4(g).. Calculate the enthalpy of combustion of ethene at 298 K and one atmosphere. Given theenthalpy of formation of CO2(g), H2O (g) and C2H4 (8) are -393.7, .... The standard enthalpy of combustion of ethene gas, C2H4(g), is -1411.1 kJ/mol at 298 K. Given the following enthalpies of formation, calculate ΔHf° for C2H4(g). dc39a6609b tcs-hr-case-study
